Australian researchers have created the most detailed cellular atlas of prostate cancer to date, identifying early molecular changes that are invisible to current pathology-based diagnostics. Published today in Cancer Research, the study also reports the discovery of a previously unknown cell type that may play a role in the development of prostate cancer.
“We’ve captured a molecular readout showing years of cancer development,” said Alexander Swarbrick, PhD, co-director of the Cancer Ecosystems Program at the Garvan Institute of Medical Research and senior author of the study. “What’s particularly surprising is the significant number of cells that look perfectly ordinary but have already acquired DNA changes associated with cancer. This suggests that current diagnostic methods using conventional microscopy may not be granular enough to detect these early mutations.”
Patient outcomes in prostate cancer are highly variable, ranging from indolent cancers that do not pose any immediate threat to rapidly advancing metastatic disease. Current prognostic methods rely on a pathologist’s analysis of biopsy samples, focusing on visual and structural differences between cancer and healthy cells. However, the study revealed that many healthy-looking cells already harbor cancer-driving genomic alterations.
“Currently we don’t know how prostate cancer starts,” said Anthony Joshua, MBBS, PhD, professor and head of the department of medical oncology at St Vincent’s Hospital Sydney. “Understanding the sequence of events that lead to life-threatening disease—and finding the signatures of cancer in normal-looking cells—is essential for creating better predictive tests and therapies.”
The researchers analyzed biopsy samples from 24 patients who had been recently diagnosed with early-stage prostate cancer. All samples were obtained from the Garvan St Vincent’s Prostate Cancer Biobank, which stores material from over 16,000 patients. Single-cell RNA sequencing and spatial transcriptomics were used to examine both prostate cancer cells and their surroundings to create a comprehensive picture of their molecular make-up and spatial distribution.
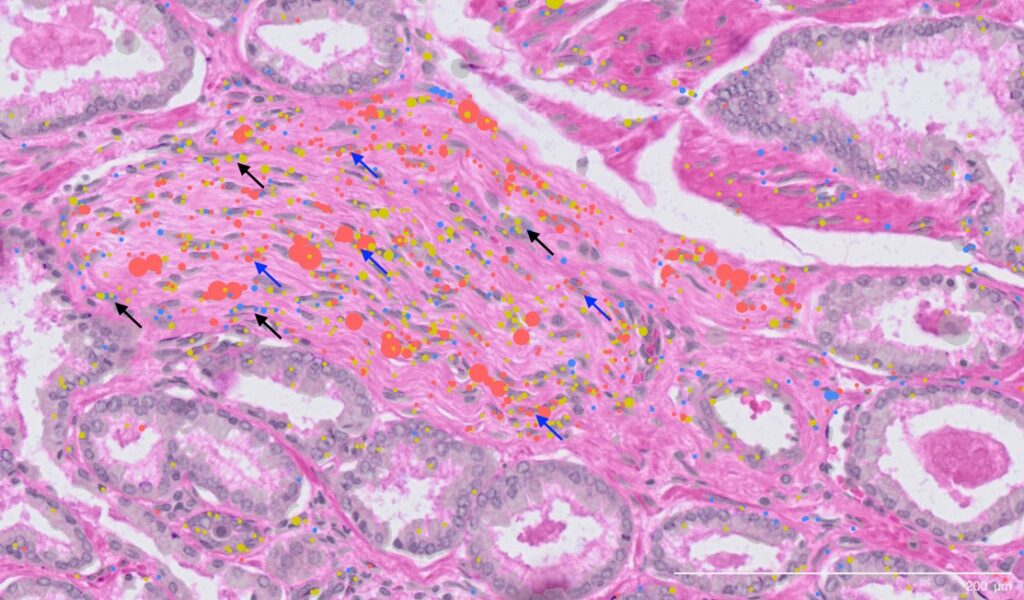
A total of 11 major cell types, 50 minor subtypes, and numerous cell states were identified, some of them unique to each individual and some shared among patients. This led to the identification of a brand new cell population that the scientists named perineural cancer-associated fibroblasts, which were located near nerves. Since prostate tumors that grow in and around nerves are associated with poorer outcomes, this discovery could provide a starting point to better understand their role in prostate cancer metastasis.
“We’ve been able to classify these cells at the molecular level for the first time, giving them a distinct identity,” said Swarbrick. “These perineural fibroblasts in tumors appear to have specialized machinery to communicate with nerves. We don’t yet know what these cells are doing, but the role of nerves in tumors is getting increasing attention in the cancer field.”
Taken together, these findings highlight how current diagnostics overlook key changes happening at the molecular and cellular levels, supporting the use of molecular testing to detect prostate cancer while it is still in the early stages.
Given the significant heterogeneity seen among prostate cancer patients, the results also provide a foundation to understanding the biological and clinical implications of the cellular and molecular make-up of each individual tumor. Future work will focus on expanding the number of patients studied and further investigating the newly identified cell type as well as the molecular changes driving cancer in healthy-looking cells.
“While most prostate cancer patients respond well to current treatments, this atlas provides foundational knowledge of the disease’s first stages that could help us pinpoint those at high risk of dangerous progression, who need more intensive intervention,” said Joshua.