Sponsored content brought to you by
Potency assays that fail during tech transfer or comparability studies don’t just delay timelines—they erode regulatory confidence and cost development cycles. Advancing drug candidates and maintaining product quality post-approval requires functional evidence of a drug’s activity, potency, and mechanism of action (MoA) using validated assays for QC and lot release to ensure batch-to-batch consistency, safety, and compliance.
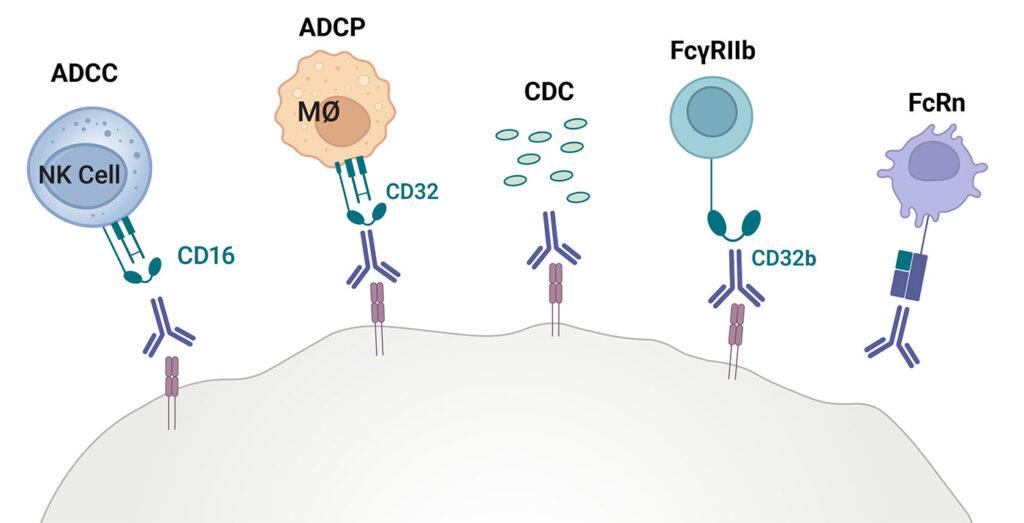
Effective against a variety of diseases, therapeutic antibodies and Fc fusion proteins have exquisite specificity in binding to an antigen, and the ability to activate an immune response through Fc effector functions like antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP).

Promega’s comprehensive assay suite provides a coherent workflow that tracks antibody Fc effector activity from binding to function. To provide greater control and confidence in Fc analytics, the bioluminescent assays adhere to ICH Q2(R2) guidelines and robustly and efficiently measure Fc effector functions across antibody development pipelines from early-stage discovery through commercial production and lot release.
The assays combine biological relevance with reproducibility while eliminating labor-intensive protocols, are scalable, and deliver decision-ready data that meet regulatory guidelines. The suite includes Lumit® FcγR Binding Immunoassays, Lumit C1q Binding Assay for CDC (Complement-Dependent Cytotoxicity), Fc Effector Reporter Bioassays for ADCC and ADCP and HiBiT Target Cell Killing Bioassays using MoA-qualified primary cells.
Screening and Ranking Candidates
During the early antibody discovery process, Lumit® FcγR Binding Immunoassays provide fast, reliable data to screen candidate binding kinetics and rank order IgG isotypes based on their affinity to the different Fcγ receptors on immune effector cells.
The add-mix-read immunoassays include all major Fcγ receptor subclasses known to be present on human effector cells including FcγRI, FcγRIIa, FcγRIIb, FcγRIIIa, and FcγRIIIb, as well as FcRn. The suite includes the allelic variants for FcγRIIa-H131 and -R131 and FcγRIIIa- V158 and -F158. Together, these receptors cover phagocytic, cytotoxic, and inhibitory immune functions.
These assays generate a luminescent readout with results available in < 60 min without immobilization artifacts, enabling 96- to 384-well high-throughput primary screens or orthogonal confirmation.
To accurately detect specificity and sensitivity across antibody variants through CDC, the Lumit® C1q Binding Assay is based on a split luciferase technology—half of the luciferase (LgBiT) is bound to C1q and half is bound to anti-IgG Fab tracer (SmBiT). A luminescent signal is generated only when the labelled C1q and an anti-IgG Fab tracer are brought into close vicinity by the test antibody, forming an active NanoBiT luciferase through complement binding.
Measuring Receptor Activation
Functional Fc Effector Reporter Bioassays directly measure Fcγ receptor activation. They employ thaw-and-use target and reporter cells expressing individual human FcγRs to provide ICH-compliant precision, accuracy, and linearity for identifying Fc binding changes that translate into functional potency shifts.
When co-cultured with an IgG antibody, the FcγR is activated and triggers promoter-driven luminescence that is detected using the Bio-Glo™ Luciferase Assay System. The add-mix-read format standardizes reagents to eliminate donor-to-donor variability and reduce background signals.
The ADCC Bioassay reflects the MoA and specificity of antibodies designed to bind and activate FcγRIIIa while demonstrating appropriate isotype specificity. The ADCP Bioassay reflects the MoA and specificity of antibodies designed to bind and activate FcγRIIa.
HiBiT Target Cell Killing Bioassays can be used to measure cytotoxic target cell killing induced by a variety of drug modalities, including validation of Fc effector function. The HiBiT TCK Bioassay pairs MoA-qualified thaw-and-use primary effector cells, including PBMCs for ADCC and macrophages for ADCP activity to reduce variability.
The bioassay features a complementation luciferase technology, where upon killing of the target cell, the HiBiT fusion protein is released into the media and binds extracellular LgBiT to create a functional luciferase enzyme. Cell death of the target cell confirms primary PBMC or macrophage activity, providing quantitative, no-wash cytotoxicity readouts that bridge binding and reporter potency data to physiologic cell death.
The integrated suite of assays provides a coherent workflow that tracks antibody Fc effector activity from initial screening through lot release. Accelerate IND and BLA filings with assays that arrive validation-ready—and stay compliant from first candidate to final lot release. Assays contain system-suitability controls, defined acceptance criteria, and lot-specific documentation, facilitating incorporation into comparability, stability, and release protocols.

Learn more www.promega.com.