ReVision Implant gets FDA Breakthrough Device nod for neural interface designed to restore vision by directly stimulating the visual cortex.
Hot on the heels of Science Corp landing a massive funding round to commercialize its retinal implant technology, Belgian neurotech ReVision Implant today announced it has received Breakthrough Device designation from the US FDA for its visual cortical prosthesis. The designation provides the company with a pathway for closer regulatory interaction as it prepares for human trials of its bionic eye technology.
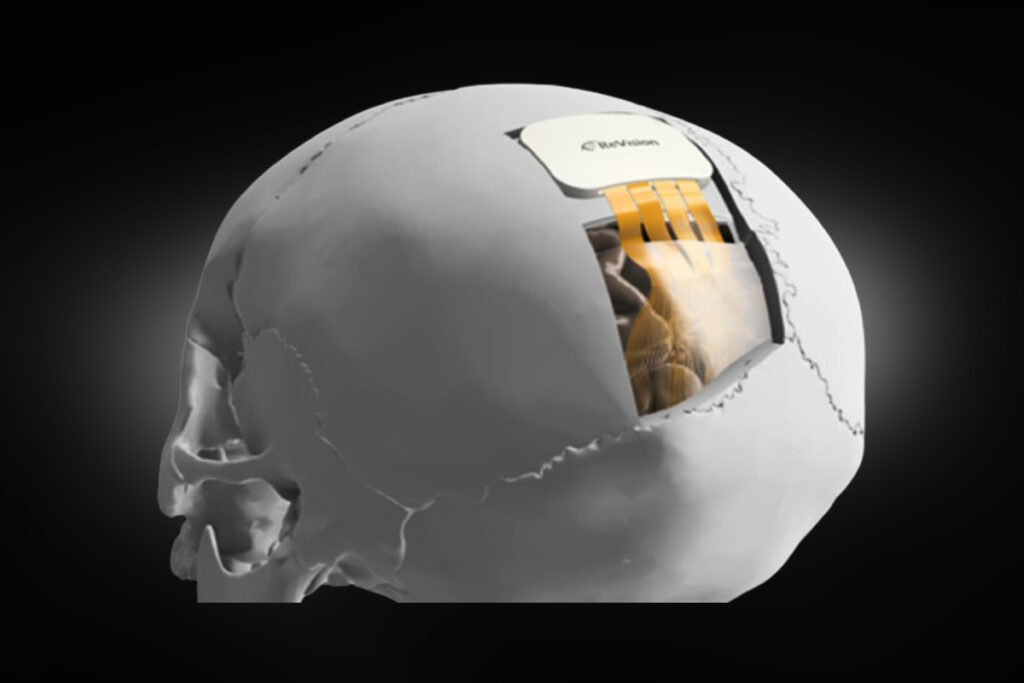
Designed to restore functional vision for people with “severe blindness” by interfacing directly with the brain, ReVision’s Occular system is aimed at patients whose vision loss cannot be addressed through retinal implants or therapies targeting the optic nerve. Rather than stimulating cells in the retina, the system delivers electrical signals directly to the visual cortex, the part of the brain responsible for processing visual information.
ReVision Implant was founded by KU Leuven university researchers Frederik Ceyssens and Peter Janssen, whose work focused on the neurophysiology of the visual cortex, brain stimulation and microfabrication of flexible implants.
“While cochlear implants have transformed treatment for hearing loss, there is still no widely available neuroprosthetic solution for restoring vision,” said Ceyssens. “Our aim is to provide people living with severe blindness with functional vision that improves independence and quality of life.”
The Occular system combines a wearable and an implanted component. A miniature camera mounted on a wireless headset captures images from the surrounding environment. These visual signals are processed and transmitted to a cortical implant containing dense arrays of microelectrodes. The electrodes stimulate neurons in patterns that the brain interprets as points of light known as phosphenes. When multiple stimulation points are activated together, they can form simple visual patterns that may allow users to identify objects or navigate through spaces.

ReVision’s approach builds on this phenomenon by distributing stimulation across a large number of electrodes to create more complex visual patterns. The company believes that increasing electrode density and refining stimulation algorithms can significantly improve the clarity of artificial vision compared with earlier prosthetic systems.
ReVision has developed an implant architecture based on ultra-flexible microelectrode arrays. Each electrode is designed to be extremely thin and biocompatible, allowing it to move with surrounding brain tissue and potentially reduce inflammation or scar formation over time. The company claims its flexible arrays can scale to hundreds or potentially thousands of electrodes distributed across regions of the visual cortex responsible for both central and peripheral vision.

Ceyssens told us how Occular differs from retinal implant approaches, which require much of the retina to still be intact.
“These people typically have lost central vision but still have peripheral vision so they’re not completely blind,” he said. “Occular, on the other hand, is more invasive as it’s a brain implant but it bypasses the optic nerve and the retina completely. Therefore, it will be useful to help in almost all cases of blindness at the cost of being more invasive. It’s mainly targeted at the types of blindness that occur at a much younger age.”
According to ReVision, extensive preclinical testing has been conducted in mice and non-human primates, including long-term studies lasting more than two years. The company claims these experiments demonstrated consistent activation of visual cortex regions through patterned electrical stimulation and showed that subjects could differentiate between stimulation patterns, suggesting the potential to convey visual information through the system.

Ceyssens told us the company has also generated some limited human data through a research collaboration with UMH university in Spain.
“Three blind volunteers were already implanted with a small electrode array, and got some vision restored, allowing better navigation, object localization and the ability to recognize some shapes such as letters,” he said. “As these implants only could cover about 1% of the visual cortex and thus restored only a few degrees of the visual field, the performance was still very limited.”
“Our main goal is to scale this up to a much larger percentage of the visual cortex using our own electrode arrays which are specifically developed to allow interfacing with a large part of the primary visual cortex, and show a proportionally better performance, obtaining a complete human proof of concept of the technology.”
The Breakthrough Device designation from the FDA will allow ReVision to work more closely with the regulator as it advances toward clinical trials. The company says it is planning an initial short-term clinical study during a scheduled brain surgery later this year to test aspects of the implant in humans. If regulatory approvals are obtained, it then aims to launch early-stage trials involving blind volunteers in the summer of 2027.
Photographs courtesy of ReVision Implant.